The Protein Metabolism Research Lab at McMaster University recognizes the importance of equity, diversity, inclusion and indigeneity (EDII) efforts within our lab group and in welcoming and promoting members of black, Indigenous and people of colour (BIPOC) and other equity-seeking communities in our research endeavours and collaborations. We support all EDII efforts. For more information, visit our Departmental statement and here for McMaster’s commitment to EDII.
The maintenance of a metabolically active skeletal muscle mass is to a great extent underappreciated, particularly where optimal health is concerned.
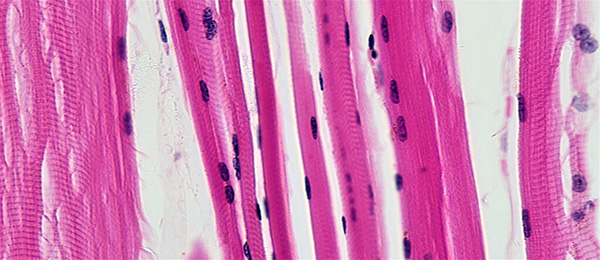
Skeletal muscle, besides its obvious role in locomotion, is a highly important thermogenic (i.e., energy consuming) tissue and the prime determinant of our basal metabolic rate, which for most of us is the largest single contributor to daily energy expenditure. Hence, declines in skeletal muscle mass can lead to increases in body fat mass. Because of its oxidative capacity (i.e., mitochondrial content) skeletal muscle is also a large site of fat oxidation, potentially playing a role in maintaining lipoprotein (cholesterol) and triglyceride homeostasis. Skeletal muscle is also, mostly by virtue of its mass, the primary site of blood glucose disposal; hence, maintaining skeletal muscle mass would also play a role in reducing risk for development of type II diabetes. Finally, the decline in maximal aerobic capacity with age, and with other muscular wasting conditions, including weight loss, has also been found to be due, to a large degree, to a decline in skeletal muscle mass and skeletal muscle quality.
Our research program has at its centre the following research question, what factors serve to maintain, increase, or decrease skeletal muscle mass? In addition, our research does not only address the absolute mass of skeletal muscle, but also its quality as assessed by the quantity of force it can generate, but also by the metabolic activity of various enzymes and energy consuming pathways.
We use a human model of resistance or aerobic exercise, immobilization, or aging to study the processes that govern: muscle accretion, in the case of resistance exercise; atrophy, in the case of immobilization; and sarcopenia, in the case of aging. In addition, my research group has studied the interaction of feeding different protein composition and varied meal timing on the processes regulating hypertrophy and disuse atrophy.
We employ stable isotope tracers of amino acids to metabolically trace the fate of ingested proteins. Muscle biopsies provide us with mechanistic information regarding processes that regulate protein accretion and degradation. We use Western blotting, RT-PCR, histological, and immunohistochemical methods to examine these mechanisms.
Information Box Group
Undergraduate Student Research Positions Protein Metabolism Research Lab Student Application
Undergraduate students interested in thesis, research practicum, or volunteer placements within the Protein Metabolism Research Lab can apply at the following link.
Applications will be reviewed by Dr. Stuart Phillips, who will contact the applicant via email.